- Home
- Weddings
- Portraits
- Journal
- Contact
- Jeff denert white pages california
- Shutter encoder download
- Defense zone 2 level 16 walkthrough
- 8gb ram mac mini 2011
- Freestyle libre flash glucose monitoring system patches
- Kanye west my beautiful dark twisted fantasy lp
- Pure hearts rescue tampa fl
- Aha moment gif
- Enable js console coderunner
- Freestyle libre flash glucose monitoring system patches skin#
- Freestyle libre flash glucose monitoring system patches full#
- Freestyle libre flash glucose monitoring system patches pro#
The potential impact on complications, is yet to be realized. Further randomized studies to assess the long-term impact on HbA1c, particularly in those with high baseline HbA1c and in specific age groups, such as adolescents and young adults, are warranted.
Freestyle libre flash glucose monitoring system patches full#
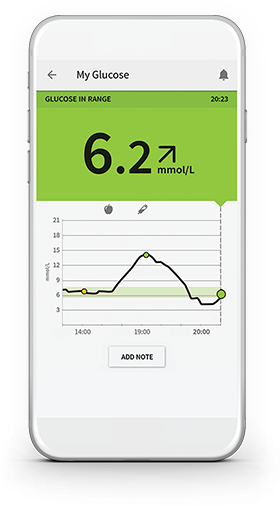
There is a need for appropriate education, of both users and healthcare professionals, to harness the full benefits. Glucose data can be visualized in multiple devices and platforms, and summarized in an ambulatory glucose profile to aid pattern recognition and insulin dose adjustment. Accuracy of the FreeStyle Libre is comparable to currently available real-time continuous glucose monitors in adults, children and during pregnancy the cost of the FreeStyle Libre is lower. User satisfaction is high and adverse events are low. In randomized trials, FreeStyle Libre use is associated with a reduction in hypoglycaemia and, in observational studies, improvements in HbA1c levels. This review discusses clinical data from randomized and observational studies, considers device accuracy metrics and deliberates its popularity and the potential challenges that this new device brings to diabetes care in the UK. Calibration free, the FreeStyle Libre is a disc worn on the arm for 14 days which is designed largely to replace the recommended 4–10 painful finger-stick blood glucose tests required each day for the self-management of diabetes. Actual results may vary among users.The FreeStyle Libre flash glucose monitor became available on prescription (subject to local health authority approval) in all four nations of the UK from November 2017, a watershed moment in the history of diabetes care. Customer reviews reflect the individual reviewer's results and experiences only and are not verified or endorsed by.
Customer reviews are provided for informational purposes only. We are not liable for inaccuracies or misstatements about products. You should use our site as a reference, carefully read all product packaging, and contact the manufacturer with any questions before using a product. While we work extremely hard to ensure that all product information is correct, manufacturers sometimes change their logos, packaging and products. Information and statements regarding dietary supplements have not been evaluated by the Food and Drug Administration and are not intended to diagnose, treat, cure, or prevent any disease. If you have or suspect that you have a medical problem, immediately contact your health care provider. You should not use the information contained herein for diagnosing or treating a health problem or disease, or prescribing medication. It is not meant to substitute the advice provided by one's physician or any other medical professional. If you order with expedited shipping please make a note that ETA will start once orders are shipped.Ĭontent and information on is provided for informational purposes only.
Freestyle libre flash glucose monitoring system patches pro#
Libre 2 sensors are intended to use with Libre 2 Reader, Not for use with Libre or Libre PRO readers.In a study by Abbott Diabetes Care, 91.1% of patients surveyed (n=123) agreed that the sensor was small and easy to wear. FreeStyle Libre 2 sensor is designed to improve patient experience.

Freestyle libre flash glucose monitoring system patches skin#
VERY THIN FILAMENT: Filament is WORN UP TO 14 DAYS ON BACK OF UPPER ARM:Scan to activate, then wait 1 hour for first glucose reading.FACTORY CALIBRATED:Accuracy not dependent on patient fingerstick calibration.WATER RESISTANT: Patients can exercise, shower and swim in a depth of up to 1 meter (3 feet) for 30 minutes.Patients scan the sensor to see their glucose reading.The systems are intended for single patient use and require a prescription. FreeStyle Libre 2, 14 day Flash Glucose Monitoring systems are continuous glucose monitoring devices indicated for replacing blood glucose testing and detecting trends and tracking patterns aiding in the detection of episodes of hyperglycemia and hypoglycemia, facilitating both acute and long-term therapy adjustments in persons (age 18 and older) with diabetes.
